- Kristian's Biology Blogs!
-
Write-ups!
- Rate of reaction in enzymes
- Aseptic Technique
- Growth curve experiment
- Model of the lungs
- Locust Dissection
- Heart dissection
- Surface area:volume practical
- Temperature and membranes
- Modelling Natural Selection
- Investgating photosynthesis using immobilised algae
- Investigating factors affecting the rate of aerobic respiration
-
Revision / Practice questions
- Explain how an organism’s abiotic environment can be affected by ecological succession.
- Explain how energy and nutrients enter, move through, and exit a food chain in an ecosystem.
- Ecologists sometimes investigate one population and at other times do research into a whole community. Define the terms population and community.
- About
- Blog
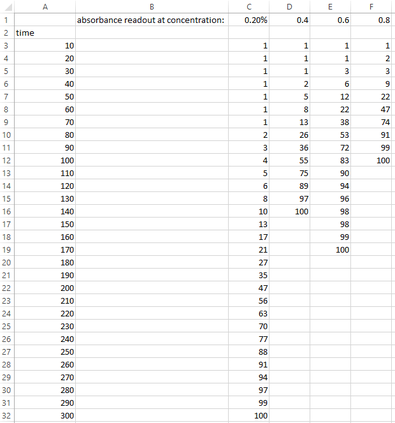
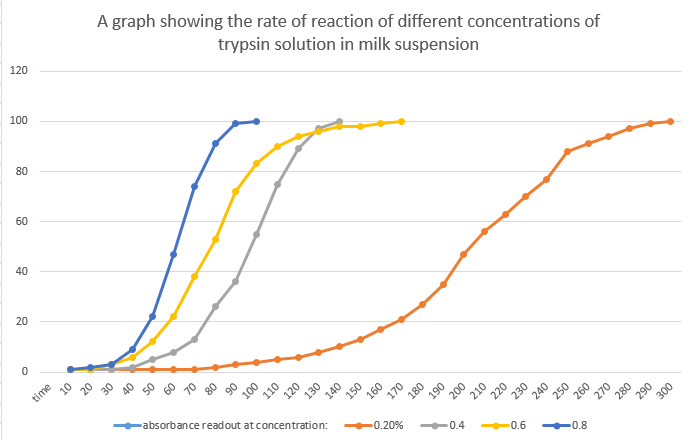
Last week, my class and I participated in an experiment investigating how the concentration of an enzyme effects the rate of reaction. This is a write up of the safety precautions taken, the equipment used, the method we used and the results we gathered, along with some questions set as homework which I will answer here. Above are the results we recorded for the rate of reactions, which clearly shows that a higher concentration of enzyme will react faster with the milk suspension. Above are my results plotted on a graph, showing a distinct pattern in concentration and time taken to fully react with the milk suspension. There does happen to be one anomaly, however, where the 0.6% solution reacted slower than the 0.4% solution. This could due to a calibration error, or possibly a miscalculation when measuring out solution.
1. What were the independent and dependent variables of this experiment? The dependent variable was the time taken for the absorbance readout to be 100% (no light absorbed) and the independent variable was the concentration of trypsin in the solution. 2. Why is it important to measure the initial rate of reaction rather than an average rate over a long time period? The initial rate of reaction will not account for there being not enough milk for the trypsin to react with, therefore it is the most accurate time to record the rate of reaction. The overall rate of reaction will show to be much slower as there may not be enough milk left to react with the trypsin toward the end, slowing down the process. 3. If the surface of the cuvette is scratched, it can result in a greater absorbance of light. If the cuvette used for the reaction was scratched (but the reference cuvette was not), would this give a random or a systematic error? why? It would be a systematic error because the error stems from damaged measuring instruments. 4. Suggest two variables that would normally be controlled in enzyme-catalysed reactions but have not been specifically controlled in this investigation. Explain why they would usually be carefully controlled and how this could be done. The two variables that were not controlled that should have been were temperature and pH of the milk solution. Both of these would dramatically affect the rate of reaction. In order to coutner temperature, you could carry the experiment out in a warmed room or store the cuvette in a water bath, and use a thermometer to ensure the temperature is stable. To control pH, you could test the pH of each solution and ensure they were similar to ensure this did not affect the rate of reaction.
1 Comment
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
